
We have a busy year in front of us; just for starters: we will be at the BSG Conference in June, we anticipate running our popular Roadshow again through 2018 and, for those of you that use our Webtool and PMS systems for IBD data collection, we are preparing for the release later this year of a completely new version of the CIMS Infoflex software that powers both of these. A wide range of people have worked together to shape the new design, and the IBD Registry team are eagerly looking forwards to sharing the first release when it is ready. We’ll keep you closely informed on developments in this as we move forwards; please do keep us closely informed with your feedback and thoughts.
In another corner, even as I write this, I know that the quarterly reports (including the Biologics Quality Account Audit) are being finalised, and I am looking forwards to those going out to participating Trusts this month. We will also be inviting Trusts to participate in the 2018-19 Audit; please do get in touch if you want to take part.
There is such huge potential in the IBD Registry. If my first two weeks are anything to go by, it’s going to be a busy few years ahead! I look forwards to being on that journey with you all, towards better IBD outcomes through better information.
Data and reporting
Quarterly reports released
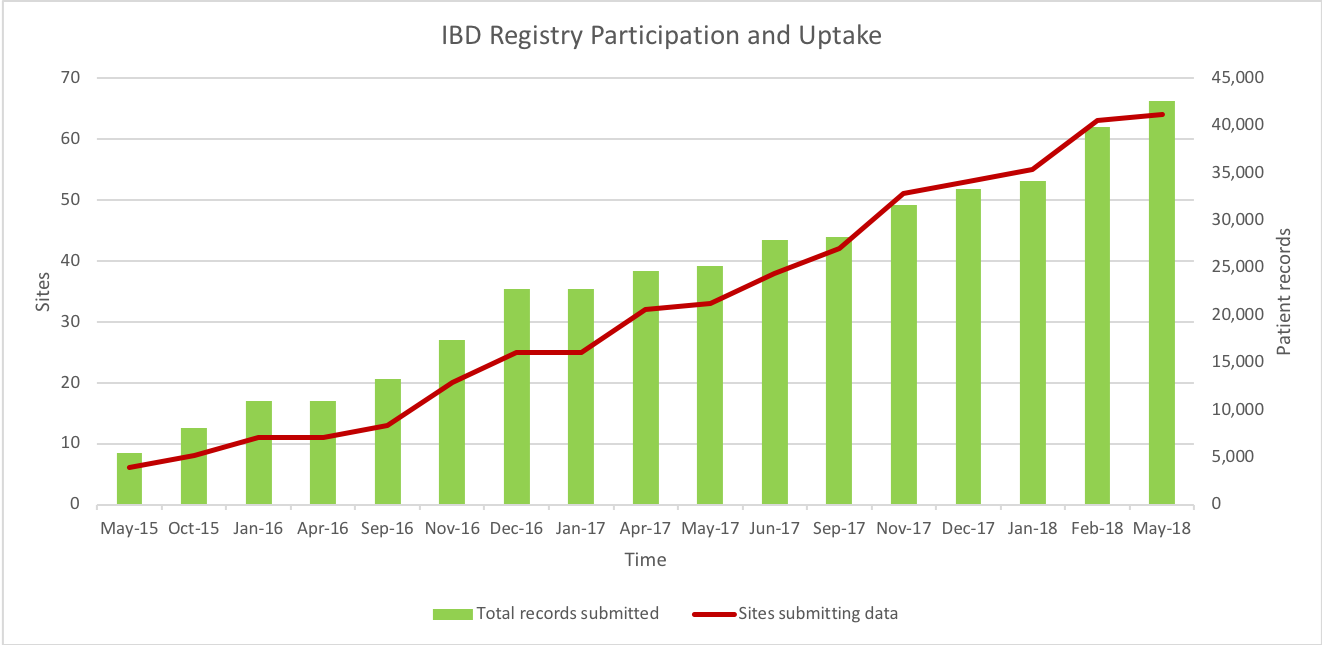
The second iteration of IBD Registry quarterly reports will be released this month. These reports will include data from the September and February submissions. Encouragingly, these show an increase of 10,000 patient records since the last quarterly report, as well as increasing depth of patient level detail, particularly for the Biologics Audit patient cohort. As a direct consequence of this, the reports now include an extended range of biologic data fields including the number of patients on individual drugs, concomitant immunosuppression, time from diagnosis to initiation of biologic therapy and disease activity indices. Site performance against the Biologics Audit will be separated into the September and February submission, allowing you to track your site’s progress.
Your comments and suggestions ([email protected]) are essential so that we can continue to make sure the reports meet your needs. Keep them coming – we really value your feedback.
IBD Biologics Audit and Quality Accounts
The IBD Registry’s IBD Biologics Audit has been included in NHS England’s list of Quality Accounts for 2018/19. As the audit is no longer nationally funded, the IBD Registry is obliged to make a small charge to participating sites to cover the costs of data handling, reporting and administration. This year the cost is £2,500, with a 20% discount for early subscribers. We’ve heard how much time it saves sites having this data ready for reporting, and we really think that the fee offers great value for money. The IBD Registry will be in touch later this month with details of how to subscribe to the IBD Biologics Audit for 2018/19.
Sites and patient numbers update
IBD Biologics Audit data submission deadlines
Here are the data submission deadlines for the IBD Biologics Audit during 2018 and 2019. You can find these on our website too. We are here to support you ([email protected]) with any issues that may arise in uploading data to NHS Digital, but please help us by allowing a bit of time to upload before the submission is due!

Information for sites
Calling all Ferring database users – deadline approaching
As many of you are aware, the development of the UK IBD Registry, with its own data entry systems, has prompted a re-evaluation of Ferring’s involvement in the provision of the IBD Database as it effectively duplicates those provided by the Registry. Ferring have decided therefore to redirect their resources and will end their sponsorship of the IBD Database from 30th June 2018. It will be possible for sites to transfer their data from Ferring to the IBD Registry.
To facilitate the transfer of data, Ferring have invested, in conjunction with North Bristol NHS Trust, in the development of Version 4. This will allow data to be uploaded to the IBD Registry (via NHS Digital), and is available to all current users of the Ferring Database. Ferring are pleased to be able to offer support to upgrade to Version 4 up to the end of June 2018.
To move to Version 4 and enable migration of your data into the UK IBD Registry, please contact Bryan Archer on 07818 027477 or [email protected]
Other news
BSG Symposium
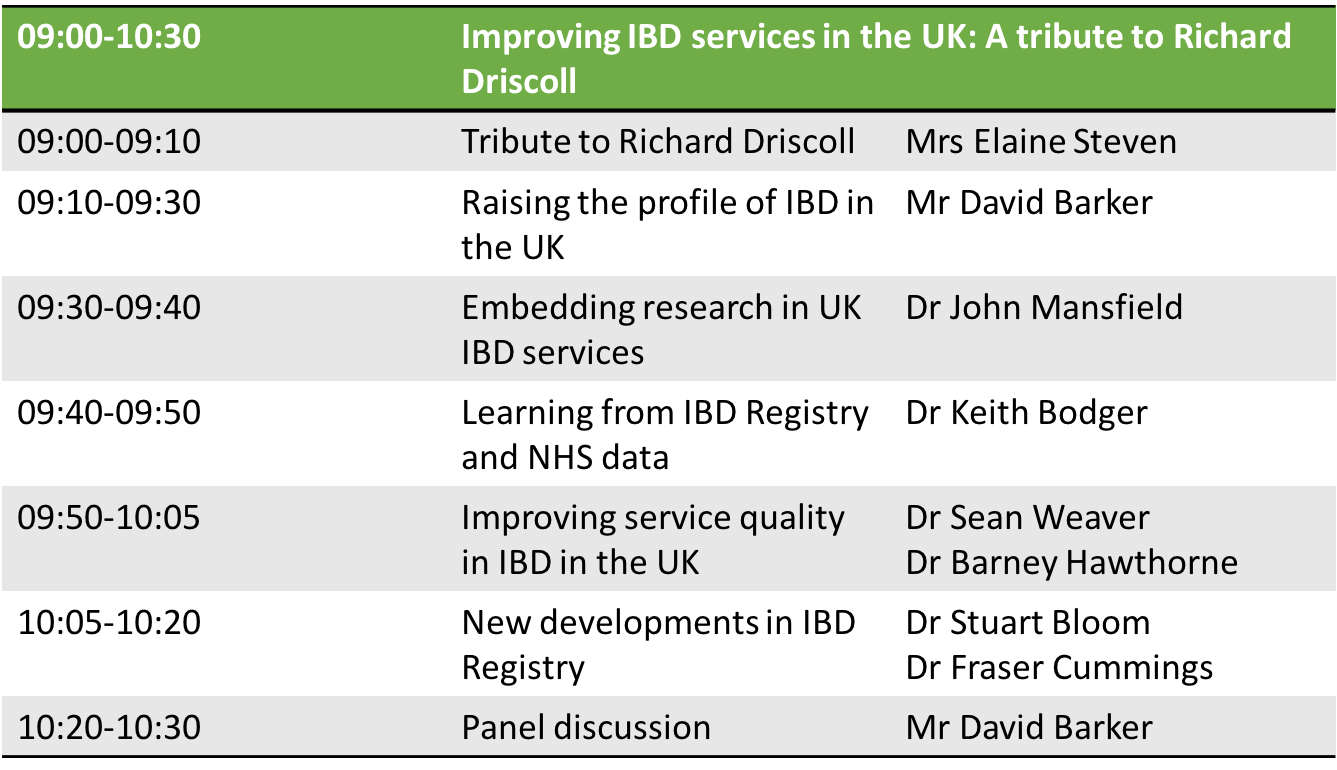
The IBD symposium at the BSG annual meeting will be held as a tribute to Richard Driscoll. ‘Improving IBD services in the UK’ will be held in Hall 3a from 9-10:30 am on Wednesday 6th June 2018. Liz will be there, along with our clinician speakers (below) and some of the team, and we hope to meet you there, to hear your views on the role of the IBD Registry in supporting the IBD community’s goals.
Newsletter consent
Do you still want to be on this mailing list?
On the 25th May 2018, the General Data Protection Regulation (GDPR) will bring in new data privacy measures. To be compliant, we’ll need your written consent to hold your email address. If you want to receive future communications from the IBD Registry team, please click this link and press send. Alternatively copy the text below and send to [email protected]
“I wish to continue receiving updates from the IBD Registry.”
If you’ve already sent an email you don’t need to do anything. If you don’t respond before the 25th May 2018 we’ll have to take you off our contact database to ensure our compliance with the new GDPR. Please note, this is just for the monthly newsletter.
With our thanks for your continued contribution to the IBD Registry,
